Detergents are amphipathic compounds with a nonpolar, hydrophobic tail and a polar, hydrophilic head group. Due to these structural features detergents tend to aggregate into structures called micelles at high enough concentration; arranging themselves with their hydrophobic tails pointed inwards and their hydrophilic heads pointed outwards. Detergents come in three types: ionic (cationic and anionic) and non-ionic. Non-ionic detergents aren’t generally used for gel electrophoresis due to their limited ability to break non-covalent interactions between protein residues and inability to impart a uniform charge onto the protein. Ionic detergents (typically anionic SDS) are used for gel electrophoresis as they are highly useful for protein solubilization, linearization and for establishing a uniform charge in preparation for gel electrophoresis.
Topics: Protein Purification, Western Blotting, Protein Estimation, Detergents, Sample Clean Up, Protein Concentration, Protein Fractionation, Protein Labeling, Protein Extraction, Protein Detection
The Use of Spectral Scanning in Nucleic Acid Purity Assessment
Spectrophotometric analysis, also called spectral scanning, of biomolecules serves two main purposes – the quantitation of nucleic acids (DNA and RNA) and purity assessment. Since the amount or concentration and purity of the DNA or RNA in a solution significantly affect the results of subsequent downstream applications, establishing these values early on can reduce, if not totally eliminate, the risk of committing errors and guarantee optimum results.
Topics: Molecular Biology, Apoptosis Assays, Assay Development (ELISA), Detergents, Buffers & Chemicals, Cytology & Histology
The Role of Tween 80 in Protein Solubilization and Stabilization
Various types of commercial-grade detergents contain elevated levels of sulfhydryl oxidizing agents, peroxides, salts, and carbonyl compounds. The proteins that are isolated by these detergents are highly susceptible to contaminating peroxides and carbonyls. The peroxides oxidize the proteins, and the carbonyl groups will form Schiff’s bases with the proteins that interfere with its structure.
Topics: Detergents
What’s the Difference Between Tween 20 and Tween 80? | G-Biosciences
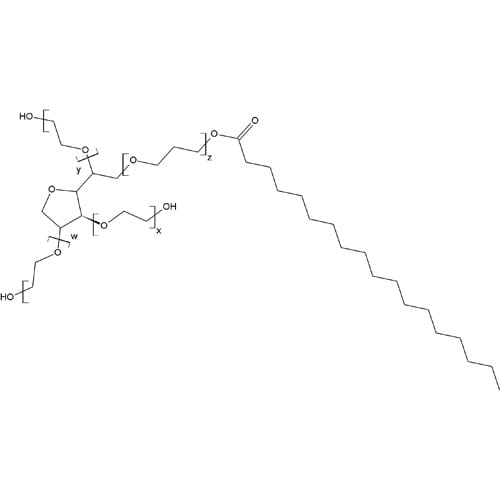
As a non-ionic viscous liquid, tweens are a class of emulsifiers used in pharmaceuticals and food preparation.
Topics: Detergents