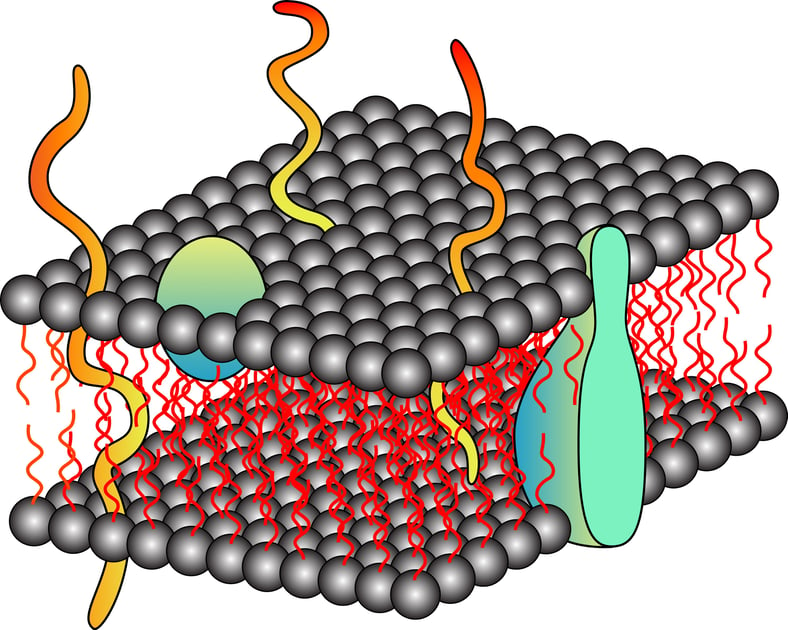
Proteins are the building blocks of cells and are the most abundant and diverse biomolecules. Proteins are literally responsible for almost all the functional aspect of a cell, from making the cellular membrane to catalyzing a biological reaction. As per the database, approximately 20-30% of the total encoded proteins have integral membrane proteins while 10-20% portion comprises membrane-associated proteins. This diverse group of proteins includes signal transducers, transporters, membrane channels, and cell surface receptors. This specialized group of proteins has become a target for therapeutic drug development in the past few decades. However, due to the unavailability of complete structural details of these proteins, targeting is a little difficult and that makes drug designing trickier than expected. The right approach is to design polished methods that can help the extraction of membrane proteins and their analysis.
Topics: Protein Purification, Detergents, Protein Extraction
The stability of proteins is crucial in many in vitro protein studies and is considered a major requirement in functional studies involving native and recombinant proteins. Thus, understanding protein stability and preserving the native conformation and normal functions of your protein of interest can be very helpful when working with your protein of interest.
Topics: Protein Purification, Protein Extraction
Expression of recombinant proteins with peptide or protein tags is widely used in protein research for three main reasons, ease of purification from a large pool of host proteins, enhancing solubility of the protein and for localization studies. Some important steps to be considered while choosing an expression vector are compatibility of tag sequence with that of the desired protein, codon usage, including linker sequences, peptide cleavage sites and the impact of the tag on the nature of desired protein. Various tags are used ranging from large proteins (Maltose Binding Protein {MBP}) to small peptides (Hexa (6X) Histidine). Tags can be added to either N-terminal side or C-terminal side of the desired protein.
Topics: Protein Purification, Molecular Biology
The Basics of Affinity Purification/Affinity Chromatography
Affinity purification, also called affinity chromatography, is a laboratory technique used for purifying protein or protein complexes within a biochemical mixture. Unlike other chromatography-based purification methods which separate molecules based on size (i.e., gel filtration or size-exclusion chromatography) or strength of ionic interaction with a solid phase material (i.e., ion exchange chromatography), affinity purification works by manipulating certain molecular properties and specific binding interactions between molecules to purify the protein of interest.
Topics: Protein Purification

.jpg?width=788&name=Protein%20Structure%20(8).jpg)




