There is a plethora of uses for detergents, both in the lab and in everyday life. The usefulness of these cleaning compounds comes from subtle differences in their chemical structure. Detergents are composed of amphipathic molecules, containing a polar hydrophilic head group attached to a long-chain hydrophobic carbon tail. The composition and charge of the molecules’ head and tail groups determines the mechanism by which these molecules will act as detergents. Because of this, different types of detergents are useful for a variety of purposes.
 IONIC
IONIC
The polar head group of ionic detergents contain either a positive (cationic) or negative (anionic) charge. Anionic detergents typically have negatively-charged sulfate groups as the hydrophilic head; whereas cationic detergents contain a positively-charged ammonium group. They are frequently used for common household soaps and cleansers, but can also serve specific purposes in a lab setting. Ionic detergents have stronger effects than uncharged detergents, because they bind to protein molecules, altering the protein’s charge and structure. They should be used when the preferred outcome involves modifying proteins and disrupting cellular structures, such as in gel electrophoresis.
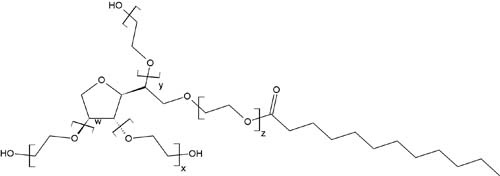 NON-IONIC
NON-IONIC
Non-ionic detergents contain molecules with head groups that are uncharged. They can be further classified into two types: polyoxyethylene (and related detergents), and glycosidic compounds. Detergents based on polyoxyethylene and similar compounds contain a neutral, polar head group and the tails are composed of hydrophobic oxyethylene chains, or, in some cases, ethylene glycoether chains. Detergents with a glycosidic base tend to use a sugar as the head group, such as glucose or maltose, and contain an alkyl polymer tail. Non-ionic detergents are less harsh than ionic detergents, having a limited ability to break protein-protein interactions. Because of this, they are considered to be non denaturing and are great for processes where keeping protein structure intact is important. These detergents are effective at isolating active membrane proteins, or breaking lipid-lipid and lipid-protein interactions.
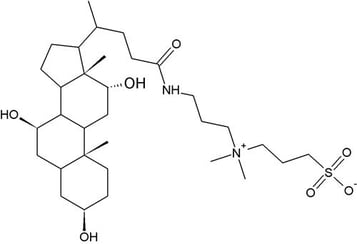 ZWITTERIONIC
ZWITTERIONIC
The polar head groups of zwitterionic detergents contain both negatively and positively charged atomic groups, therefore the overall charge is neutral. The strength of action of these compounds is considered to be intermediate between ionic and non-ionic detergents, sharing characteristics with both types. Similar to non ionic detergents, zwitterionic detergents have a neutral net charge. However, they are often more efficient at breaking protein-protein bonds, similar to ionic detergents, but the extent to which proteins are affected is less harsh. Zwitterionic detergents effectively break the protein bonds while maintaining the native state and charge of the individual proteins. The versatility of zwitterionic detergents are useful for a variety of purposes, such as: chromatography; different types of electrophoresis, including 2D gel electrophoresis; mass spectrometry; and solubilization of organelles and inclusion bodies.
When deciding which detergent to use, the following should be taken into account:
- pH, ionic charges, and other factors that could affect the detergent or target molecules
- the desired extent of how target molecules will be affected (ex. Completely denatured or undenatured)
- the desired end result, including structure and charge.






